|
10/6/2023 0 Comments Kcl lattice energy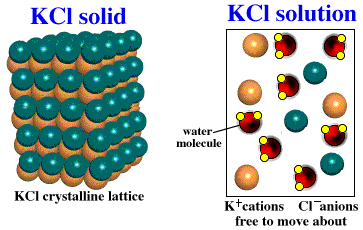
This effect is illustrated in Figure 4.2.2, which shows that lattice energy decreases for the series LiX, NaX, and KX as the radius of X − increases. For example, the calculated value of U for NaF is 910 kJ/mol, whereas U for MgO (containing Mg 2 + and O 2− ions) is 3795 kJ/mol.īecause lattice energy is inversely related to the internuclear distance, it is also inversely proportional to the size of the ions. Source: Data from CRC Handbook of Chemistry and Physics (2004).īecause the lattice energy depends on the product of the charges of the ions, a salt having a metal cation with a +2 charge (M 2 +) and a nonmetal anion with a −2 charge (X 2−) will have a lattice energy four times greater than one with M + and X −, assuming the ions are of comparable size (and have similar internuclear distances). 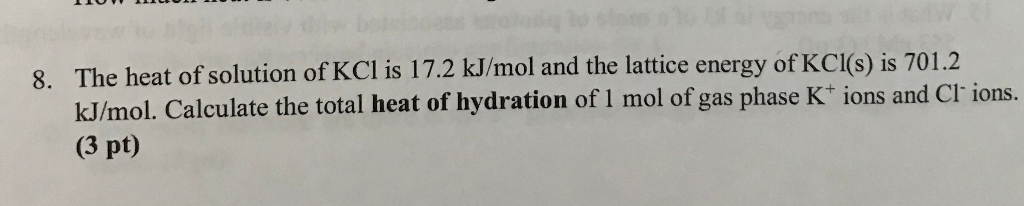
Note that r 0 may differ between the gas-phase dimer and the lattice. In such an arrangement each cation in the lattice is surrounded by more than one anion (typically four, six, or eight) and vice versa, so it is more stable than a system consisting of separate pairs of ions, in which there is only one cation–anion interaction in each pair. While Equation 4.1.1 has demonstrated that the formation of ion pairs from isolated ions releases large amounts of energy, even more energy is released when these ion pairs condense to form an ordered three-dimensional array. These properties result from the regular arrangement of the ions in the crystalline lattice and from the strong electrostatic attractive forces between ions with opposite charges. They are not easily deformed, and they melt at relatively high temperatures. Ionic compounds are usually rigid, brittle, crystalline substances with flat surfaces that intersect at characteristic angles. Metal ores are commonly combinations of metal atoms with oxygen atoms, and this combination is produced when metals rust, so the process where electrons are transferred to the oxygen atoms from the metal atoms is known as oxidation of the metal and the reverse process, where pure metals are produced is called reduction of the ore to the metal. The reaction of a metal with a nonmetal usually produces an ionic compound that is, electrons are transferred from the metal to the nonmetal. 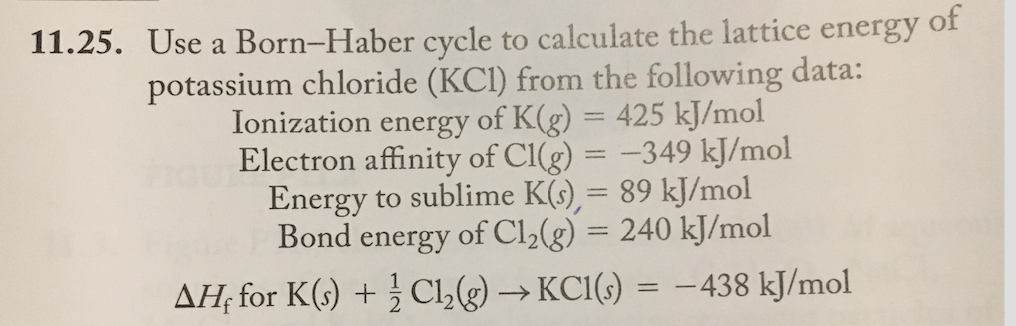
To understand the relationship between the lattice energy and physical properties of an ionic compound.higher ionization of lithium is more than compensated by the higher lattice energy and pushing the equilibrium as potassium vapor partial pressure is being decreased by vapor condensation.\( \newcommand\) Thermodynamic parameters are shown in the table below. 
With potassium vapors being eliminated by condensation on colder surfaces of the apparatus, used in the video. That is because higher ionization energy is more than compensated by more negative hydration enthalpy of $\ce$$ But lithium has more negative standard redox potential (which applies in water context only). Potassium reacts more violently with water than lithium. TNT is much more reactive than coal, but released specific energy during coal burning is 10 times bigger than during TNT explosion. Reactivity (kinetics) and tendency of reaction outcome (thermodynamics) are independent. Note that element electronegativity $\chi$ is a combined indicative quantity from covalent bond context, so its comparative values do not always apply in scenario analysis what happens or should be preferred.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
